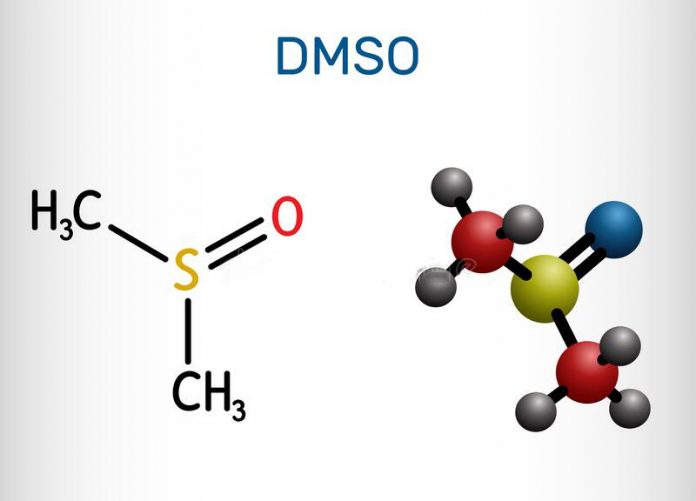
DMSO (dimethyl sulfoxide) is a colorless liquid with the chemical formula (CH3)2SO. It was first described by a Russian chemist, Alexander Saytzeff, in 1866, as a byproduct of paper manufacturing.
For nearly a century after its discovery, DMSO remained little more than a curiosity. In the 1950s, however, the chemical found increasing industrial use as a versatile solvent and antifreeze.

Due to its striking ability to penetrate human skin and tissues and enter the bloodstream—and to its apparent anti-inflammatory properties—DMSO also acquired a cult reputation for healing a variety of ills, including arthritis, burns, sprains, skin ulcers, and autoimmune conditions. Since DMSO has never been readily available as a pharmaceutical, most users acquired preparations that were manufactured for industrial use.
By 1965, approximately 100,000 Americans were using DMSO, mainly for the treatment of minor burns, sprains, bruises, and arthritis. Then, in November 1965, a Scottish woman who applied DMSO in conjunction with several medications abruptly died from an allergic reaction.
This fatality, along with evidence from laboratory studies that DMSO caused damage to the lenses of the eyes of animals, prompted the FDA to ban further clinical tests on the substance. Oddly, though, only a year later these restrictions were eased to allow investigations of DMSO’s utility in conditions like interstitial cystitis, scleroderma, and rheumatoid arthritis.
Now, over 50 years later, more than 50,000 papers on the chemistry and clinical implications of DMSO have been published. In the United States, DMSO is FDA-approved only as a preservative for organs for transplant, for veterinary use, and for treating interstitial cystitis in humans. DMSO is also extensively used in medically-related technologies, such as polymerase chain reaction analysis and cryo-storage.
While it is generally agreed that—when used properly—DMSO is safe (Gaylord Chemical Company, DMSO Health and Safety Information, Bulletin 106), there is little else about this molecule that doesn’t generate debate.

Current Evidence for Medical Use of DMSO
- Interstitial cystitis: Fair evidence exists for the use of DMSO. Indeed, DMSO may work when other treatments fail. Benefits of treatment seem to outweigh the risks.
- Amyloidosis, diabetic ulcers, gastritis, herpes zoster (shingles), increased intracranial pressure (e.g., following injury or due to tumor or infection), reflex sympathetic dystrophy, rheumatoid arthritis, tendinitis: At least fair scientific evidence exists for benefits, but benefits may not outweigh risks except in special cases. Not enough evidence to generally recommend DMSO therapy.
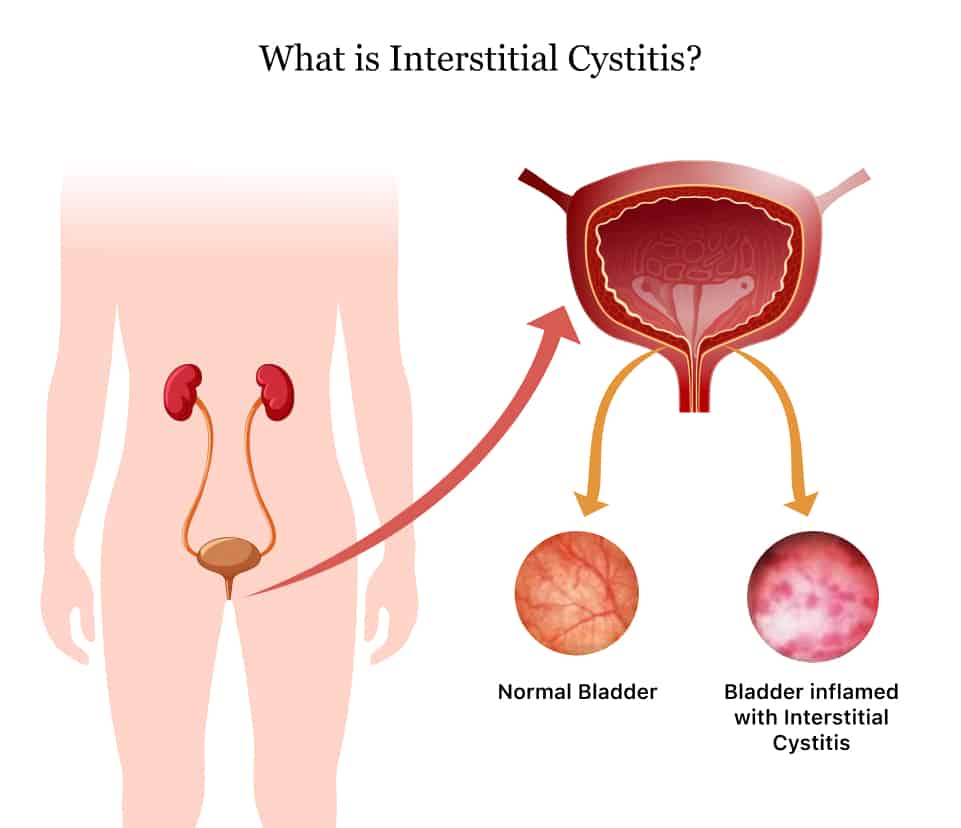
- Scleroderma: DMSO does not seem to be effective and is not recommended.
- Acute herpes, Alzheimer’s, burns, cancer, colitis, fibromyalgia, gallstones, hypercholesterolemia, muscle pain, pancreatitis, schizophrenia, lupus, tuberculosis, ulcers: No evidence of benefit. Risks may outweigh benefits. DMSO therapy is discouraged.
Given the tendency for Western physicians to anchor their clinical decisions in evidence-based medicine, and given DMSO’s status as a relatively cheap, natural substance (hence, pharmaceutical companies aren’t eager to conduct research into its applications) it may be quite some time before DMSO enjoys FDA-sanctified acceptance by the American medical establishment.
In the meantime, DMSO still has a solid base of supporters among both laypeople and health care professionals.